The following contains information that was presented as supplemental material for a poster presentation at CGU 2012. A link to the original poster can be found at the bottom of the section.
All fats are not equal: Considerations when using fatty acid biomarkers in compound-specific stable isotope (CSSI) soil and sediment tracing-Supplemental
Introduction
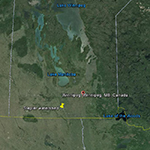
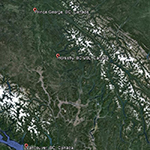
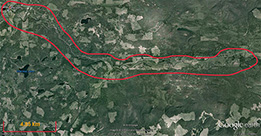
Research is currently be carried out in the South Tobacco Creek (STCW), Manitoba and Horsefly (HRW), British Columbia, Canada, watersheds. The STCW has been well-studied and monitored with a well-documented history, making this an ideal location for evaluating soil and sediment tracing techniques.
I am conducting research in the HRW and STCW under the supervision of my committee members Drs. Ellen Petticrew and Phil Owens (UNBC) and Dr. David Lobb (University of Manitoba) as part of my PhD research in order to evaluate soil and sediment tracing using compound-specific stable isotopes, primarily (13C and 12C). Although CSSIs have been successfully used in a number of areas (such as food webs) coupled with unmixing models in order to assign source nitrogen, carbon, etc., only recently has the
possibility of using CSSIs in sediment tracing been investigated. Two papers of note which have looked at this possibility are by Gibbs (2008) and Blake et al. (2012).
 |
 |
| |
Fig. 1. Overview of the STCW |
|
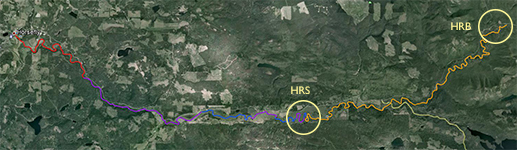
Fig. 2. Overview of the HRW |
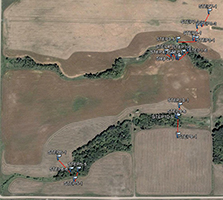
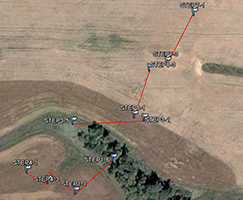
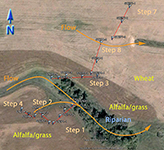
An extensive sampling regime has been established in the STCW, which is intended to address questions of natural variability. Part of the sampling regime is shown in Fig. 3. The watershed is divided into subwatersheds, and crops have been planted accordingly. For example, the wehat field and alfalfa/grass fields drain into different subwatesheds. This minimizes the mixing of CSSI signals between the two crop types as effectively as possible. This type of isolation of crops into differing subwatersheds is typical for the STCW for research purposes.
Some of the natural variability the sampling regime in the STCW is attempting to capture includes variability:
- along transects (shown in Fig. 3 as Step 1, Step 2, etc.) for a particular crop type
- between transects of the same crop type
- between transects of differing crop type
The variability the sampling regime is intended to capture includes variability due to:
- moisture regime (water use efficiency)
- orientation to the sun
- differences between crop types
- soil profile
- soil strength
Other points within the watershed and downstream from the subwatersheds in Fig. 3.are also being sampled for differences in crop type i.e. field samples as well as for transport. Sediment samples have been taken at two points downstream from the main sampling site.
 |
 |
| |
Fig. 3. Partial sampling regime in the STCW |
|
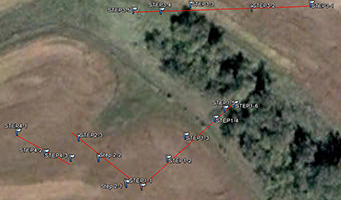
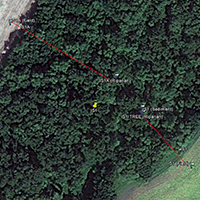
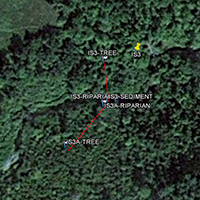
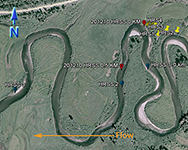
Fig. 4. Partial sampling regime in the HRW |
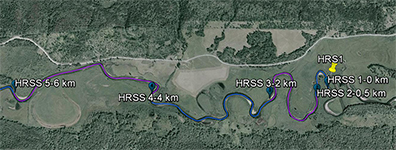
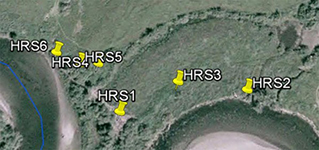
An inset of part of the sampling regime in the HRW is shown in Fig. 4. The HRW is a relatively non-diverse watershed in terms of agricultural activity; the primary agricultural activity is haying. The remainder of the land is mostly mixed temperate forest (e.g. spruce, fir, cedar, etc.). Variability in the CSSI signal due to crop difference is thus expected to be low relative to the STCW.
One of the main reasons for selecting the HRW is the size of the watershed. Resuspended sediment samples have been taken at various points downstream from the sampling sites shown in Fig. 4. at differeing intervals (e.g. 0.5, 1, 2 km, etc.). The furthest point downstream is approximately 28 km from the main sampling site. The purpose is to investigate the transport of the CSSI signal and observe changes in the signal in a large watershed.
This page will be further updated in the future. If you have any questions and wish to contact me, please click on the 'Home' tab, where my contact information can be found. Thank you for visiting my page.
Poster references
- Ainsworth, E. A., and Long, S. P. (2004) What have we learned from 15 years of free-air CO2 enrichment (FACE)? A meta-analytic review of the responses of photosynthesis, canopy properties and plant production to rising CO2, New Phytol., 165, 351-372.
- Badeck, F.-W. et al. (2005) Post-photosynthetic fractionation of stable carbon isotopes between plant organs—a widespread phenomenon, Rapid Commun. Mass Spectrom., 19, 1381-1391.
- Cernusak, L. A. et al. (2009) Why are non-photosynthetic tissues generally 13C enriched compared with leaves in C3 plants? Review and synthesis of current hypotheses, Funct. Plant Biol., 36, 199-213.
- Chikaraishi, Y., and Naraoka, H. (2003) Compound-specific δD-δ13C analyses of n-alkanes extracted from terrestrial and aquatic plants, Phytochemistry, 63, 361-371.
- Farquhar, G. D. et al. (1989) Carbon isotope discrimination and photosynthesis, Annu. Rev. Plant. Physiol. Plant. Mol. Biol., 40, 503-537.
- Glaser, B. (2005) Compound-specific stable-isotope (δ13C) analysis in soil science, J. Plant Nutr. Soil Sc., 168, 633-648.
- Hobbie, E. A., and Werner, R. A. (2004) Intramolecular, compound-specific, and bulk carbon isotope patterns in C3 and C4 plants: A review and synthesis, New Phytol., 161, 371-385.
- Jetter, R., and Kunst, L. (2008) Plant surface lipid biosynthetic pathways and their utility for metabolic engineering of waxes and hydrocarbon biofuels, Plant J., 54, 670-683.
- Körner, C. et al. (1988) A global survey of carbon isotope discrimination in plants from high altitude, Oecologia, 74, 623-632.
- Ohlrogge, J. B., and Jaworski, J. G. (1997) Regulation of fatty acid synthesis, Annu. Rev. Plant. Physiol. Plant. Mol. Biol., 48, 109-136.
- Van de Water, P. et al. (2002) Leaf δ13C variability with elevation, slope aspect, and precipitation in the southwest United States, Oecologia, 132, 332-343.
- Wang, G. et al. (2010) Altitudinal trends of leaf δ13C follow different patterns across a mountainous terrain in north China characterized by a temperate semi-humid climate, Rapid Commun. Mass Spectrom., 24, 1557-1564.
- Warren, C. R., and Adams, M. A. (2000) Water availability and branch length determine δ13C in foliage of Pinus pinaster, Tree Physiol., 20, 637 -643.
- Wei, K., and Jia, G. (2009) Soil n-alkane δ13C along a mountain slope as an integrator of altitude effect on plant species δ13C, Geophys. Res. Lett., 36, 5 PP.
- Wu, Y. et al. (2009) Effects of different soil weights, storage times and extraction methods on soil phospholipid fatty acid analyses, Geoderma, 150, 171-178.
- Xu et al. (2010) An improved method for determining medium- and
long-chain FAMEs using gas chromatography, Lipids, 45, 199-208.
A copy of the poster can be found
here. Note this poster is subject to copyright.